Medication-Related Osteonecrosis of the Jaw
Position Statement of the American College of Prosthodontists
PDF link
The American College of Prosthodontists agrees with the recommendation for the term, “Medication- Related Osteonecrosis of the Jaw (MRONJ)” suggested by the American Association of Maxillofacial Surgeons (AAOMS) to replace the previous, more-specific term, “Bisphosphonate-Related Osteonecrosis of the Jaw (BRONJ),” due to the increasing number of osteonecrosis cases resulting from the use of other antiresorptive and antiangiogenic therapies. For a comprehensive review of the pathophysiology, differential diagnosis, incidence, staging, and surgical management of MRONJ several references are suggested for review, but this topic is considered outside the scope of this position statement.1-3 The purposes of this position statement are threefold:
1.) to make recommendations for aggressive eradication of odontogenic pathosis prior to the commencement of medical treatment modalities associated with MRONJ
2.) to review the existing literature regarding the prosthodontic considerations for the patient undergoing these treatment modalities
3.) to make recommendations as to the possible oral prosthetic rehabilitation for patients who have suffered functional or cosmetic losses due to MRONJ and associated surgical treatments.
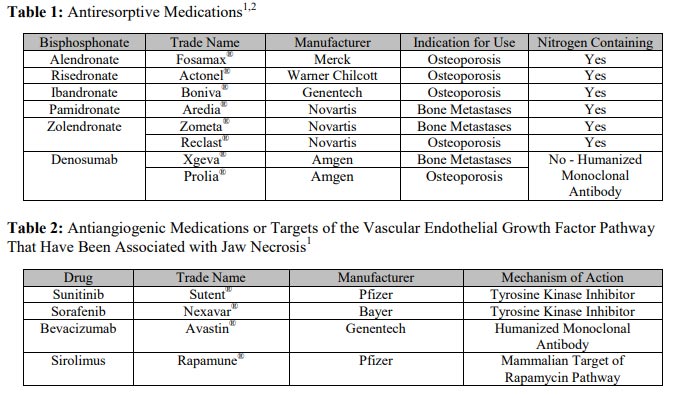
MRONJ Definition
Patients may be diagnosed with MRONJ if the following three situations are present: 1.) they are receiving or have received treatment with antiresorptive or antiangiogenic medications, 2.) they present with exposed necrotic bone in the maxillofacial region that has persisted for more than 8 weeks and 3.) they have no history of radiation therapy or diagnosed metastatic disease to the jaws.1,4,15
It is well-documented that oral bisphosphonate use for the osteoporotic patient is far less likely to lead to osteonecrosis when compared to the oncologic patient receiving intravenous bisphosphonate or antiresorptive medications.5 However, patients about to begin an oral regimen should be educated as to the potential risks of oral healing complications later in life if surgical treatment interventions are indicated. In addition, patients who have received oral bisphosphonates for 4 years or longer6 or who have other comorbidities (rheumatoid arthritis, glucocorticoid exposure, diabetes, and smoking) must be considered to have a higher risk of developing MRONJ following oral surgical procedures, including implant placement, than those patients receiving these drugs for less than 4 years.1,15 A recent publication noted that current non-evidence-based recommendations are trending toward proceeding with dental care with little or no treatment modifications for patients on IV or oral bisphosphonates for osteoporosis, and also stated that with ongoing research studies and consensus groups, it is anticipated that evidence-based recommendations are forthcoming.7
Patients who have received oral bisphosphonates for osteoporosis for less than 4 years and have no additional clinical risk factors may receive surgical procedures without delay or alteration of the procedure. In addition, restorative, prosthetic, endodontic, and periodontal procedures may be treatment planned and provided without contraindication. In this group of patients, implant placement has not been shown to have an increased risk of MRONJ development;8-10 however, it is recommended that the patient be given a specific informed consent that highlights the possibility of delayed implant failure coupled with the low risk of MRONJ development secondary to the continued use of oral bisphosphonates.
Patients who have received oral bisphosphonates for osteoporosis for less than 4 years and have additional clinical risk factors OR patients who have received oral bisphosphonates for more than 4 years with or without additional clinical risk factors may receive surgical procedures; however, based on current literature, a 2 to 3 month drug holiday should be considered for this group of patients with a higher risk of developing MRONJ.1,11 Working with the prescribing physician, the oral bisphosphonate should not be restarted until osseous healing has occurred.1 Implant placement must be carefully considered, and alternative conventional therapies given ample consideration in this group of patients.8
For patients about to begin intravenous antiresorptive therapies, a comprehensive evaluation by a prosthodontist or other oral health care provider must be completed as the first step of an effective riskreduction strategy. Appropriate dental screening and subsequent treatment prior to commencement of antiresorptive therapy has been shown to lower the risk of ONJ.12,13 The aim of this evaluation and any subsequent treatment is to treat restorable teeth and extract all teeth deemed to have a hopeless or extremely guarded prognosis prior to the onset of therapy. Given that in the majority of MRONJ cases, recent dentoalveolar trauma was the most prevalent and consistent risk factor,14,15 an aggressive management strategy must be undertaken prior to the commencement of these therapies. In addition to the elimination of acute infection and sources of potential infection that may become problematic during antiresorptive therapy such as tooth mobility, root fragments, asymptomatic periapical pathosis, and caries, the clinician must also carefully assess the patient’s motivation, education, and understanding of the importance of home oral care. In a recent study limited to breast cancer patients, it appears that dental implants that are well-integrated prior to the initiation of IV bisphosphonate therapy are not a risk factor for the development of MRONJ.16 It is clear that a larger study looking at multiple diseases and multiple drugs must be completed in order to provide evidence-based conclusions that existing implants are not a risk factor for MRONJ development. There is a clear benefit to good oral and dental health in the prevention of MRONJ,17 and consideration to the addition of daily chlorhexidine rinses or fluoride treatments to home care oral regimens should be given.18 Complete and partial denture patients must have a thorough evaluation of the retention, stability, and comfort of the prostheses. Fabrication of new removable prosthetics, if indicated, must follow strict prosthodontic guidelines so as to not lead to mucosal irritation, ulceration, and possible development of MRONJ. It also must be noted that a twofold risk for MRONJ has been reported for denture wearers receiving IV antiresorptive therapy for cancer.19 Special care and consideration must be given to the mandibular lingual flange areas of any removable prosthesis, as this is a site that has very thin mucosa and therefore has a higher likelihood of ulceration. A study revealed that patients wearing dentures had a significantly shorter duration to onset of MRONJ than patients not wearing dentures, thereby supporting the need for regular recalls and denture modifications as indicated.20
Patients receiving IV bisphosphonates or antiangiogenic drugs for cancer must receive ongoing continued recall care with a recall interval of 3, 4, or 6 months dependent upon the patient’s ability to maintain adequate home care coupled with the periodontal status of their teeth. The goal of dental or prosthodontic therapy is to minimize or eliminate the need for surgical intervention.21 Teeth deemed unrestorable should be considered for endodontic therapy, coronectomy,22 and possible root cap placement. Drug holidays are reported to be useful only in cases of oral bisphosphonate use uncomplicated by cancer, methotrexate, or prednisone.23 In this group of patients, implant placement is not recommended.
Little evidence exists at the present time to direct the prosthodontic management of patients with a history of bisphosphonate use. It is likely that patients who have active MRONJ have decreased tissue tolerance to functional loads imparted to these tissues by removable prostheses.24 Prosthetic reconstruction of the patient who has active MRONJ should be postponed until the area of active disease has been eradicated. Following adequate healing, prosthetic reconstruction can proceed with caution, giving careful consideration to areas of previous disease and those prone to additional irritation and ulceration from removable prosthetics.25-28 Those areas include the midpalatal area of the maxilla and the mylohyoid ridge areas of the mandible. Conventional fixed or removable prosthodontic rehabilitation may be treatment planned and provided; however, implant placement should be avoided. Patients who receive removable prostheses must be educated to contact their prosthodontist or restorative dentist at the first sign of irritation from the prosthesis. Routine and thorough intraoral examinations, hygiene, and periodontal maintenance procedures must be scheduled to lessen the likelihood of the development of recurrent or new areas of MRONJ for the life of the patient.
It is the position of the American College of Prosthodontists (ACP) that
1.) Patients planned to begin either oral or IV antiresorptive therapies should have a comprehensive clinical and radiographic evaluation prior to the onset of therapy followed by aggressive eradication of acute infection, advanced chronic infection, and sites of potential infection. Implants that are integrated and present without signs of peri-implant pathosis prior to the initiation of medical therapy do not require removal.
2.) Management of patients planned to begin, who have already initiated, and those who have completed antiresorptive therapies require a multidisciplinary approach to care. Identification of early-stage MRONJ is critical, and immediate referral to experienced surgical colleagues will provide the affected patient with the most predictable outcome.
3.) Implant placement in patients with a history of IV antiresorptive medications or in patients with 4 or more years of oral therapy is not without potential complications. For patients with a history of IV bisphosphonates or other antiangiogenic drugs for the treatment of cancer, implant placement is not recommended. For the patient with a greater than 4-year history of receiving oral bisphosphonate medications, careful consideration for each patient situation, including a drug holiday, must be given to develop a risk-benefit analysis prior to initiating surgical placement of dental implants.
4.) Long-term management of these patients should follow the Clinical Practice Guidelines for Recall and Maintenance of Patients with Tooth-Borne and Implant-Borne Dental Restorations published by the ACP.29
References
1. American Association of Oral and Maxillofacial Surgeons: Position paper on medication-related osteonecrosis of the jaw. Surgical Update 2015;25:2-17
2. Otto S (ed): Medication-Related Osteonecrosis of the Jaws. Berlin, Springer-Verlag, 2015
3. DePonte FS (ed): Bisphosphonates and Osteonecrosis of the Jaw: A Multidisciplinary Approach. Milan, Springer-Verlag, Italia, 2012
4. Kim KM, Rhee Y, Kwon YD, et al: Medication related osteonecrosis of the jaw: 2015 Position Statement of the Korean Society for Bone and Mineral Research and the Korean Association of Maxillofacial Surgeons. J Bone Metab 2015;22:151-165
5. Marx RE, Sawatari Y, Fortin M, et al: Bisphosphonate-induced exposed bone (osteonecrosis/osteoporosis) of the jaws: Risk factors, recognition, prevention, and treatment. J Oral Maxillofac Surg 2005;63:1567-1575
6. Lo JC, O’Ryan FS, Gordon NP, et al: Prevalence of osteonecrosis of the jaw in patients with oral bisphosphonate exposure. J Oral Maxillofac Surg 2010;68:243-253
7. Division of Reproductive and Urologic Products, Office of New Drugs Division of Pharmacovigilance II, Office of Surveillance and Epidemiology Division of Epidemiology, Office of Surveillance and Epidemiology Center for Drug Evaluation and Research Food and Drug Administration: Background Document for Meeting of Advisory Committee for Reproductive Health Drugs and Drug Safety and Risk Management Advisory Committee. September 9, 2011. Available online at http://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/drugs/DrugSafetyRiskMana gementAdvisoryCommittee/ucm27o958.pdf. (Page 19). Accessed January 2, 2016
8. Napeñas JJ, Kujan O, Arduino PG, et al: World Workshop on Oral Medicine IV: Controversies regarding dental management of medically complex patients: assessment of current recommendations. Oral Surg Oral Med Oral Pathol Oral Radiol 2015;120:207-226
9. Grant BT, Amenedo C, Freeman K, et al: Outcomes of placing dental implants in patients taking oral bisphosphonates: a review of 115 cases. J Oral Maxillofac Surg 2008;66:223-230
10. Tallarico M, Canullo L, Erta X, et al: Dental implant treatment outcomes in patient under active therapy with alendronate: 3-year follow-up results of a multicenter prospective observational study. 2015 Jul 14. doi: 10.1111/clr.12662. [Epub ahead of print]
11. Ata-Ali J, Ata-Ali F, Peñarrocha-Oltra D, et al: What is the impact of bisphosphonate therapy upon dental implant survival? A systematic review and meta-analysis. Clin Oral Implants Res 2014;25:1-9
12. Damm DD, Jones DM: Bisphosphonate-related osteonecrosis of the jaw: A potential alternative to drug holidays. Gen Dent 2013;61:33-38
13. Bonacina R, Mariani U, Villa F, et al: Preventive strategies and clinical implications for bisphosphonaterelated osteonecrosis of the jaw: A review of 282 patients. J Can Dent Assoc 2011;77;b14714. Vandone AM, Donadio M, Mozzati M, et al: Impact of dental care in the prevention of bisphosphonate-associated osteonecrosis of the jaw: A single-center clinical experience. Ann Oncol 2012; 23:193-200
14. Vandone AM, Donadio M, Mozzati M, et al: Impact of dental care in the prevention of bisphosphonateassociated osteonecrosis of the jaw: A single-center clinical experience. Ann Oncol 2012; 23:193-200
15. Gupta S, Gupta H, Mandhyan D, et al: Bisphosphonate related osteonecrosis of the jaw. Natl J Maxillofac Surg 2013;4:151-158
16. Matsuo A, Hamada H, Takahashi H, et al: Evaluation of dental implants as a risk factor for the development of bisphosphonate-related osteonecrosis of the jaw in breast cancer patients. Odontology 2015 May 9. [Epub ahead of print]
17. Kreimmel M, Ripperger J, Hairass M, et al: Does dental and oral health influence the development and course of bisphosphonate related osteonecrosis of the jaws (BRONJ)? Oral Maxillofac Surg 2014;18:213-218
18. Hinchy NV, Jayaprakash V, Rossitto RA, et al: Osteonecrosis of the jaw – prevention and treatment strategises for oral health professionals. Oral Oncol 2013;49:878-886
19. Vahtsevanos K, Kyrgidis A, Verrou E, et al: Longitudinal cohort study of risk factors in cancer patients of bisphosphonate-related osteonecrosis of the jaw. J Clin Oncol 2009;27:5356-5362
20. Hasegawa Y, Kawabe M, Kmura H: Influence of dentures in the initial occurrence site on the prognosis of bisphosphonate-related osteonecrosis of the jaws: a retrospective study. Oral Surg Oral Med Oral Pathol Oral Radiol 2012;114:318-324
21. Stewart DL: Prosthodontic treatment of a patient taking nitrogen-containing bisphosphonates to preserve the integrity of the epithelial attachment: a clinical report. J Prosthet Dent 2011;106:350-354
22. AAE Position Statement: Endodontic Implications of Bisphosphonate-Associated Osteonecrosis of the Jaws. Chicago, American Association of Endodontists, 2011
23. Marx RE: A decade of bisphosphonate bone complications: what it has taught us about bone physiology. Int J Oral Maxillofac Implants 2014;29:e247-e258
24. Saldanha S, Shenoy VK, Eachampati P, et al: Dental implications of bisphosphonate-related osteonecrosis. Gerodontology 2012;29:177-187
25. Kyrgidis A, Teleioudis Z, Vahtsevanos K: The role of the dental hygienist in the prevention of osteonecrosis of the jaw in patients wearing dentures. Int J Dent Hyg 2010;8:154
26. Kyrgidis A, Vahtsevanos K: Increased risk for bisphosphonate-related osteonecrosis of the jaws in patients wearing dentures could be attributable to impaired mucosal cell wound healing. J Oral Maxillofac Surg 2009;67:1355-1356
27. Levin L, Laviv A, Schwartz-Arad D: Denture-related osteonecrosis of the maxilla associated with oral bisphosphonate treatment. J Am Dent Assoc 2007;138:1218-1220
28. Infante-Cossio P, Lopez-Martin JC, Gonzalez-Cardero E, et al: Osteonecrosis of the maxilla associated with cancer chemotherapy in patients wearing dentures. J Oral Maxillofac Surg 2012;70:1587-1592
29. Bidra AS, Daubert DM, Garcia LT, et al: Clinical practice guidelines for recall and maintenance of patients with tooth-borne and implant borne dental restorations. J Prosthodont 2016;25:S32-S40
Authors
Mark C. Hutten, DDS, MS, FACP
Approved ACP Board of Directors: Feb. 28, 2016
Reaffirmed ACP Board of Directors Feb. 28, 2021
© Copyright 2022 American College of Prosthodontists. All rights reserved